
Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Multiply the number of calcium atoms by 3 and the number of phosphorus atoms by 2.Formula in Hill system is C 4H 6K Na O 65Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. Phosphorus goes from 0 to -3, gaining 3 electrons (oxidation).Calcium goes from 0 to +2, losing 2 electrons (reduction).Identify the changes in oxidation numbers:.In Ca 3P 2, calcium has an oxidation number of +2, and phosphorus has an oxidation number of -3.Phosphorus (P) also has an oxidation number of 0 in its elemental form.Calcium (Ca) has an oxidation number of 0 in its elemental form.Process: identify the oxidation numbers, determine the changes in oxidation state, balance the atoms that change their oxidation state, and then balance the remaining atoms and charges. Useful for redox reactions, this method involves balancing the equation based on the change in oxidation numbers.īest For: Redox reactions where electron transfer occurs. Balance the oxygen atoms by placing a coefficient of 2 in front of H 2O:. 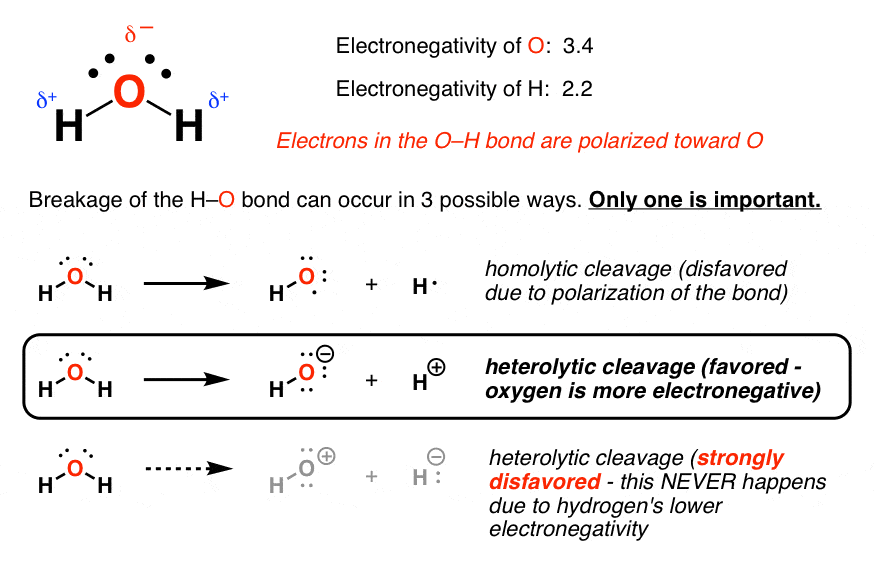
There are 2 O atoms on the left and 1 O atom on the right. There are 2 H atoms on the left and 2 H atom on the right.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
